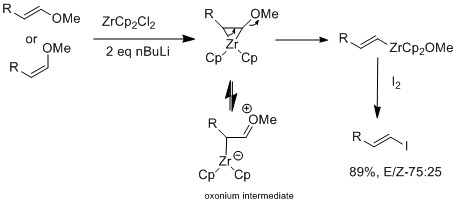
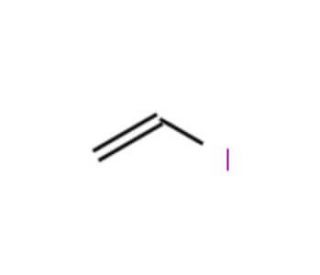
Expedient Synthesis of Large‐Ring trans‐Enamide Macrolides by CuI‐Mediated Intramolecular Coupling of Vinyl Iodide with Amide: Total Synthesis of Palmyrolide A - Yadav - 2016 - European Journal of Organic Chemistry - Wiley

HIA excess alc. KOHA CI./hy 25. Product © is (1) Alkyl iodide (3) Vinyl iodide (2) Vinyl chloride (4) Allyl chloride

An efficient cis-reduction of alkyne to alkene in the presence of a vinyl iodide: stereoselective synthesis of the C22–C31 fragment of leiodolide A - ScienceDirect