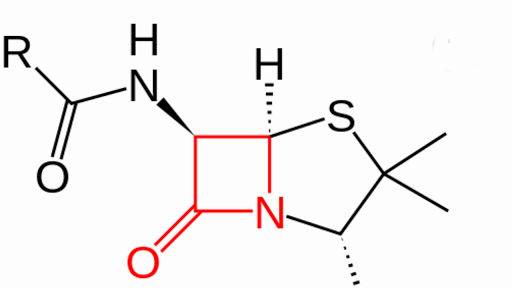
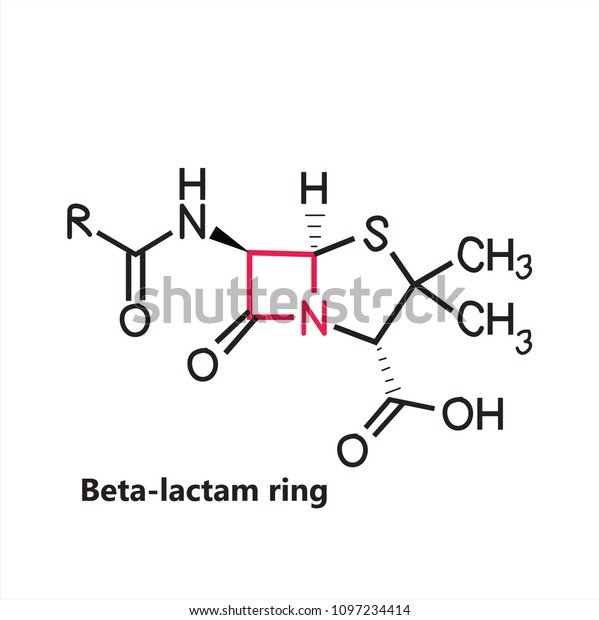
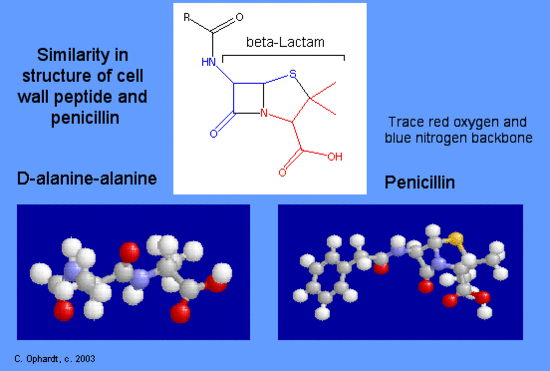
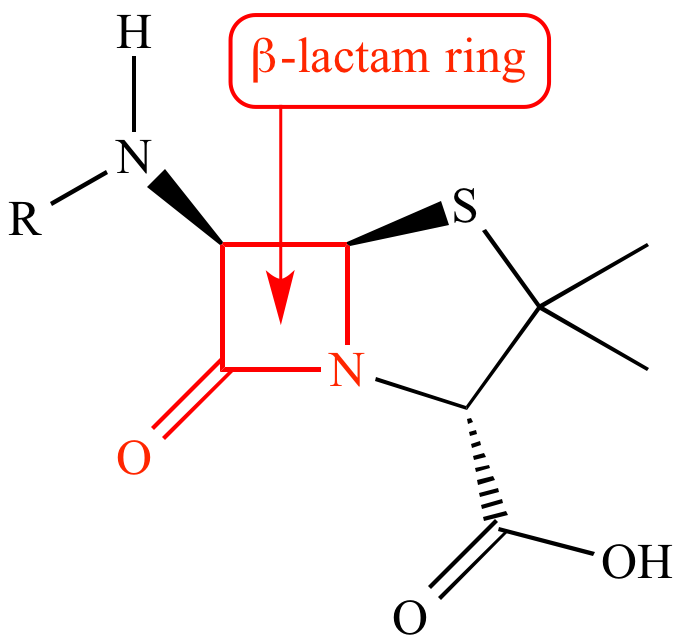
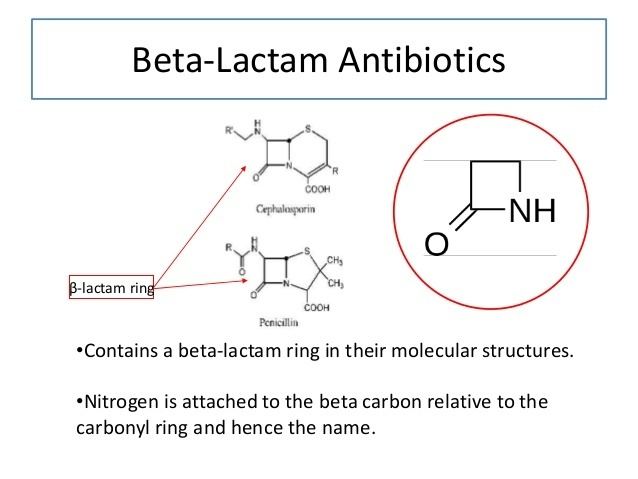
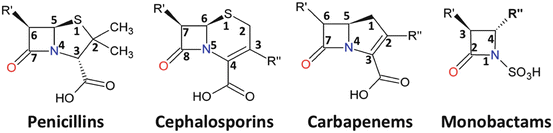
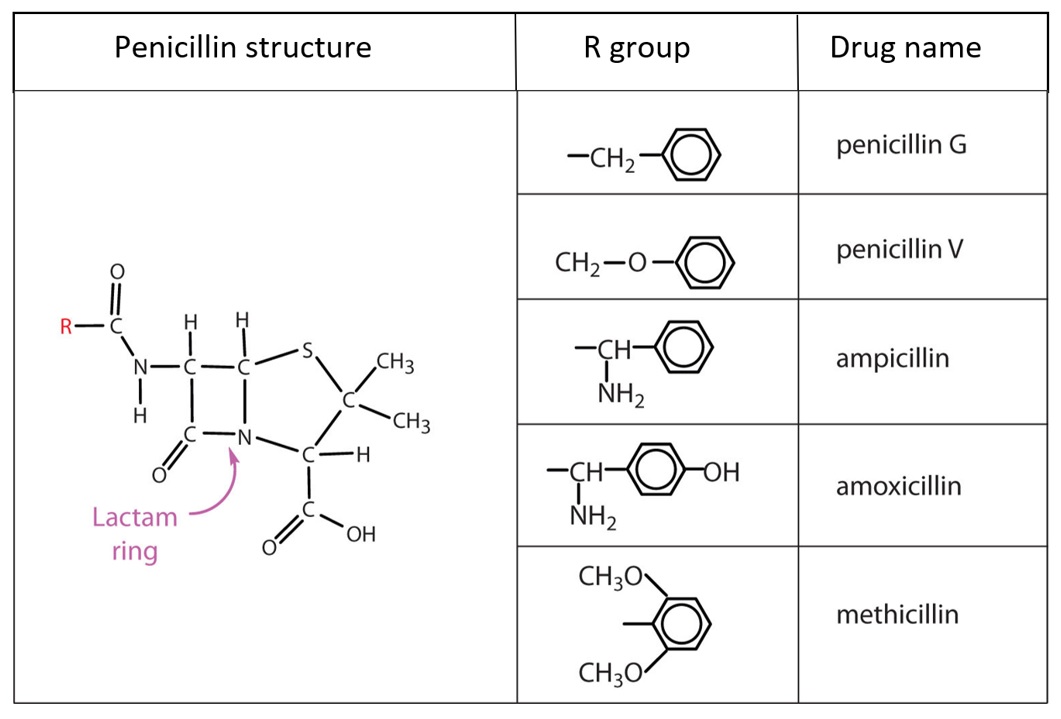
References in Cross-reactivity in β-Lactam Allergy - The Journal of Allergy and Clinical Immunology: In Practice
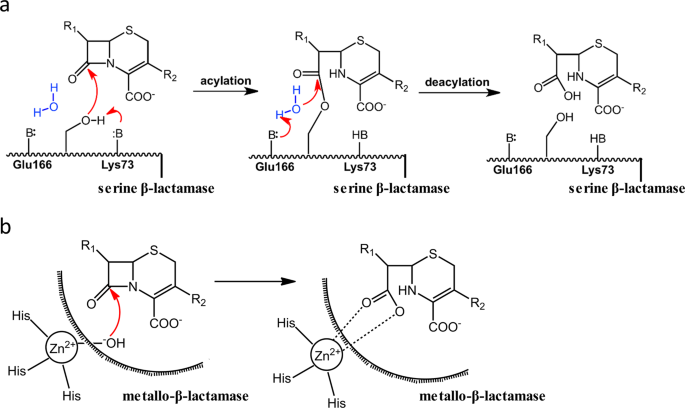
The hydrolytic water molecule of Class A β-lactamase relies on the acyl-enzyme intermediate ES* for proper coordination and catalysis | Scientific Reports

What is the function of the other components of beta-lactam antibiotics besides the beta-lactam ring? - Quora

Chemical structures of the beta-lactam antibiotics discussed in this... | Download Scientific Diagram

Modified Penicillin Molecule with Carbapenem-Like Stereochemistry Specifically Inhibits Class C β-Lactamases | Antimicrobial Agents and Chemotherapy
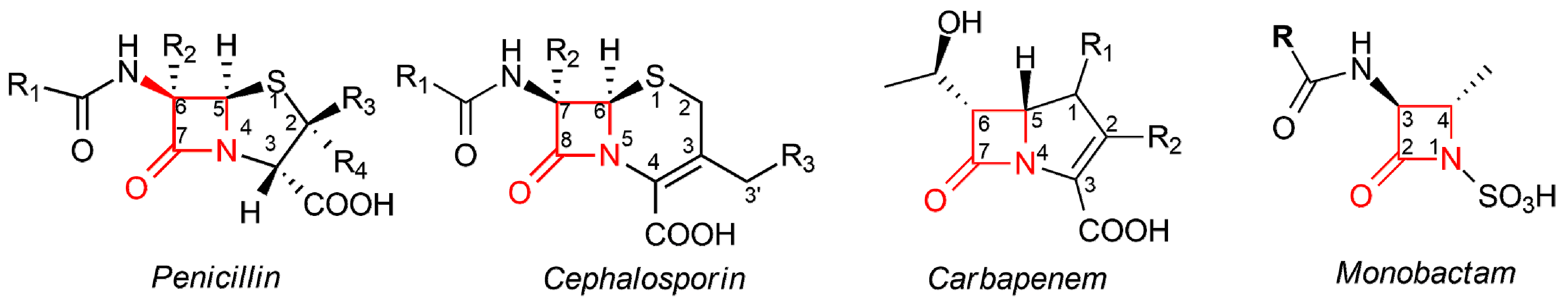
IJMS | Free Full-Text | The Odd Couple(s): An Overview of Beta-Lactam Antibiotics Bearing More Than One Pharmacophoric Group | HTML

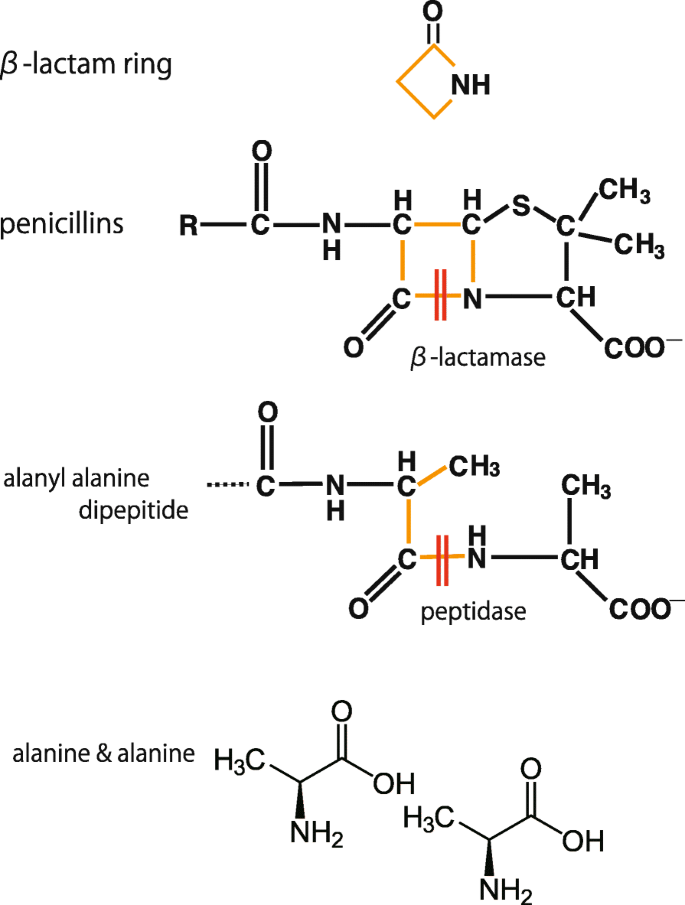
Osmosis - Penicillins are antibiotics that belong to the group of beta lactams, which have a beta-lactam ring in their structure that helps inhibit cell wall synthesis in bacteria. As a result,
Molecular diversity of extended-spectrum β-lactamases and carbapenemases, and antimicrobial resistance | Journal of Intensive Care | Full Text

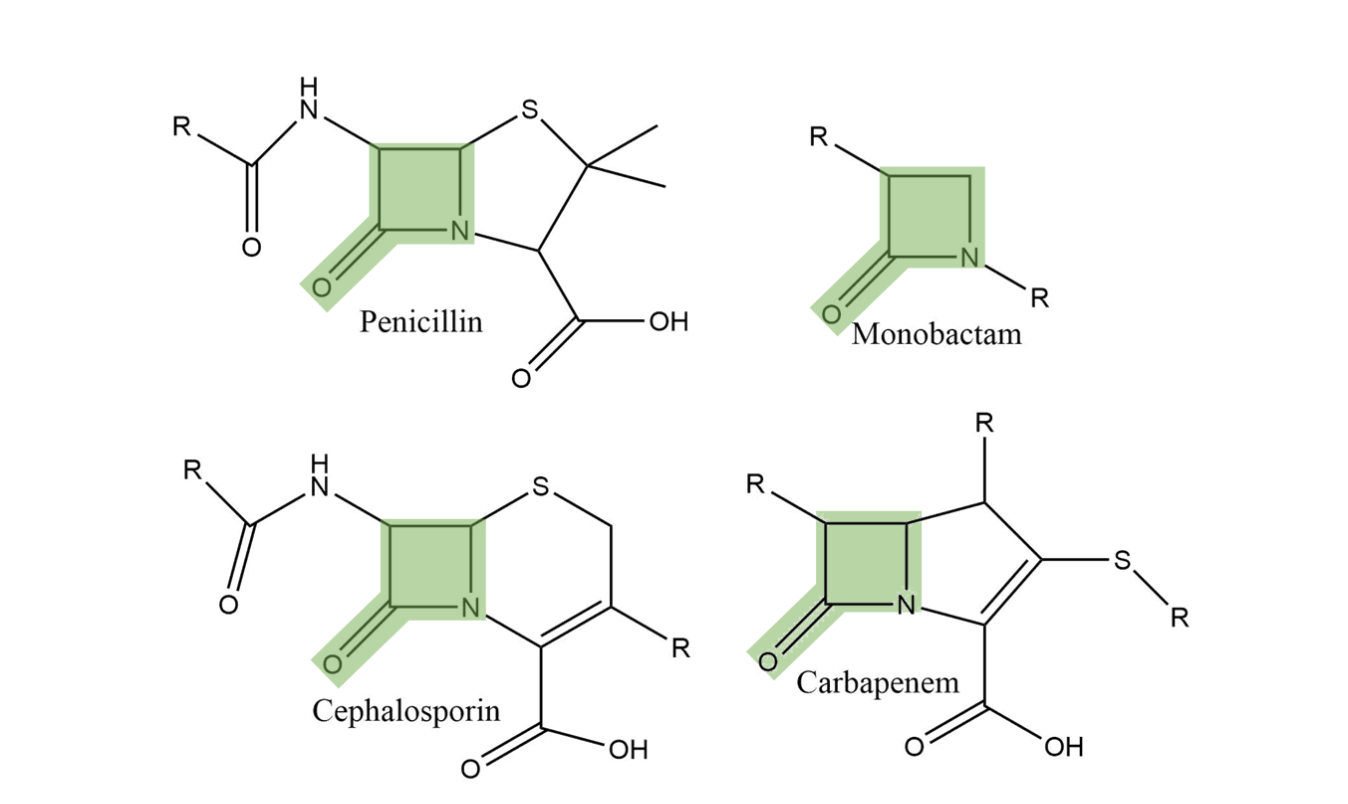

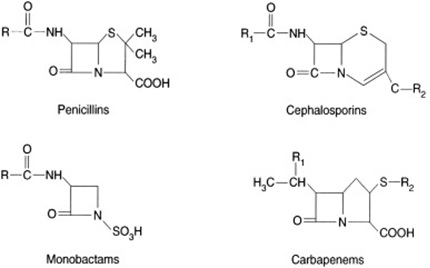
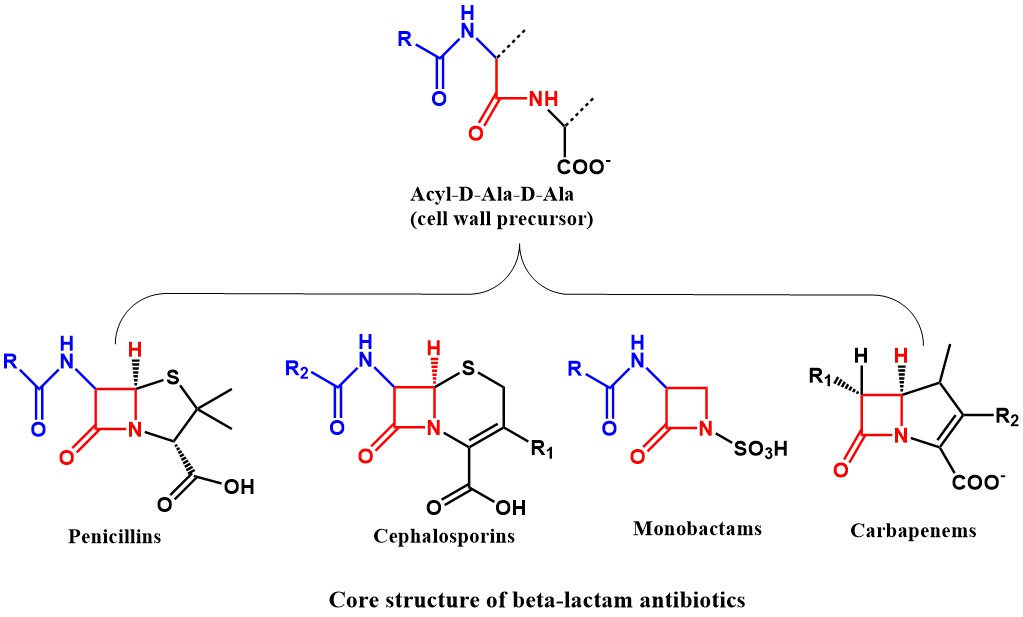
![betalactam_pharm [TUSOM | Pharmwiki] betalactam_pharm [TUSOM | Pharmwiki]](https://tmedweb.tulane.edu/pharmwiki/lib/exe/fetch.php/betalactam_structure.png?w=600&tok=54cff1)